Navigation auf uzh.ch
Navigation auf uzh.ch
Investigations into the role of the immune system in resistance to E. multilocularis has been largely limited to studies of the metacestode stage of infection, where it is known that parasite-specific humoral and cell-mediated immune responses are both progressively established. However, no studies investigating the immune parameters and factors involved in susceptibility or resistance to E. multilocularis oncosphere invasion have been carried out until our recent work on Rattus norvegicus, a host for which natural AE cases are rarely described. Our experiments first compared the susceptibility of immunocompetent Wistar rats to oral infection with eggs or oncospheres and intraperitoneal infection with oncospheres or metacestode material. Immunocompetent rats were totally resistant to oral infection, whereas they were susceptible to experimental intraperitoneal infection which led to metacestode development. This suggests that resistance of rats to oncosphere invasion after oral infection involves different mechanisms to those that control the development of AE. Our subsequent experiments included animals with an altered immunological status, such as congenital T-cell deficient nude or pharmacologically immunosuppressed Wistar rats. These experiments revealed that the absence of an adaptive (T-cell) immune response is not the source of resistance against oncosphere invasion in rats, while treatment with dexamethasone (DXM) led to establishment of the parasite in the liver. This led to our hypothesis that primarily involved innate immune mechanisms, provided by e.g., granulocytes, NK-cells and macrophages are involved in the host defense against E. multilocularis-oncosphere invasion. This hypothesis could be confirmed in our recent animal experiment, where immunocompetent rats were individually or combined depleted from the latter mentioned innate immune cell populations. Granulocyte depletion induced by polyclonal antibodies resulted in successful intestinal oncosphere invasion with subsequent metacestode development in the livers of rats. This new developed rat model is a substantial improvement for future infection experiments with oral inoculation of eggs to study AE in rats.
Institute members: Joekel Deborah, Maria Teresa Armua-Fernandez, Philipp A. Kronenberg, Peter Deplazes (Project Leader)
Funding sources: Forschungskredit der Universität Zürich
In collaboration with: Bernard Vanhove and Salomé Leibund Gut-Landmann, Section of Immunology, Institute of Virology, University of Zurich
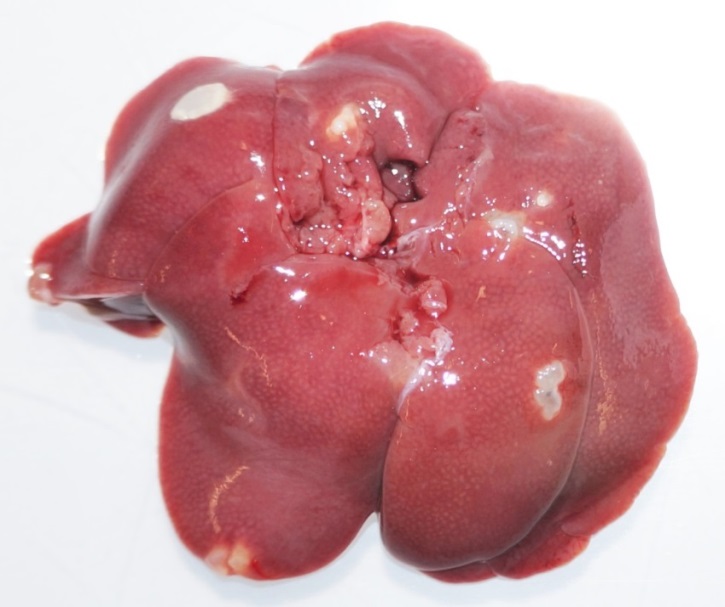
Multiple E. multilocularis metacestode development in granulocyte depleted rats, 10 weeks after oral egg inoculation.